The New Rules of Global Pharmaceutical Manufacturing Company

How Top Companies Are Rebuilding Supply Chains, Embracing AI, and Winning in 2026 By Kilitch Healthcare | Pharmaceutical Manufacturing Company Insights The World Changed. Did Your Pharmaceutical Supply Chain? Not long ago, the global pharmaceutical manufacturing company operated on a simple promise: manufacture at scale, ship globally, repeat. Then the world had other plans. Pandemic-era disruptions, geopolitical tensions, regulatory overhauls, and surging demand for specialty therapeutics forced every pharmaceutical manufacturing company to take a hard look in the mirror. The ones that survived the storm didn’t just adapt — they reinvented themselves entirely. Welcome to 2026. The rules are different now. The global pharmaceutical manufacturing company landscape has never been more competitive, complex, or — for those ready to lead — more full of opportunity. Companies that are thriving today aren’t doing so by luck. They’ve rebuilt their supply chains from the ground up, adopted AI and automation as strategic assets, and anchored everything in an unwavering commitment to quality. At Kilitch Healthcare, we live these realities every single day. And in this article, we’re pulling back the curtain on what the new rules of global pharmaceutical manufacturing actually look like in practice — and what it takes to win. Rule #1: Resilience Is No Longer Optional in Your Supply Chain For decades, the pharmaceutical supply chain ran on the principle of efficiency above all else. Single-source suppliers. Just-in-time inventory. Concentrated manufacturing hubs. It worked — until it didn’t. The disruptions of the early 2020s exposed a brutal truth: a supply chain optimized only for cost is a supply chain optimized for fragility. In 2026, top global pharmaceutical manufacturing company are rebuilding with resilience as the non-negotiable foundation. What does that look like? Geographic diversification is the new norm. Smart manufacturers are no longer concentrating production in a single country or region. India continues to assert itself as one of the world’s most critical pharmaceutical manufacturing hubs — accounting for approximately 20% of global generic medicine exports — but the emphasis now is on creating manufacturing networks that can flex, scale, and reroute when disruption hits. Dual and multi-sourcing of APIs. The days of relying on a single Active Pharmaceutical Ingredient (API) supplier are largely over for serious players. Leading global pharmaceutical manufacturing company now maintain multiple vetted API partnerships, ensuring that a single point of failure doesn’t cascade into a patient care crisis. Real-time supply chain visibility. The most progressive manufacturers have invested heavily in end-to-end digital traceability — knowing exactly where every raw material, intermediate, and finished product is at every stage. This isn’t a luxury; it’s a competitive requirement. At Kilitch Healthcare, our world-class manufacturing facility spanning over 3,50,000 sq ft is built with this resilience philosophy at its core — compliant with international standards, equipped to serve regulatory markets globally, and designed for the kind of operational continuity that global partners depend on. Rule #2: AI Isn’t the Future of Pharmaceutical Manufacturing — It’s the Present If you’re still treating Artificial Intelligence as something on the horizon for your pharmaceutical manufacturing operations, you’re already behind. In 2026, AI has moved from pilot projects and proof-of-concepts into the beating heart of manufacturing operations for the world’s leading pharmaceutical companies. The impact is transformative across every function. Predictive Quality Control. AI systems now analyze thousands of process variables in real time, flagging deviations before they become batch failures. For sterile injectables and ophthalmic formulations — categories where quality is literally life-or-death — this capability is game-changing. Rather than catching problems at the end of production, manufacturers using AI-driven quality systems catch them at the beginning or mid-stream, dramatically reducing waste, recalls, and compliance risk. Intelligent Manufacturing & Process Optimization. Machine learning algorithms continuously analyze production data to optimize parameters like temperature, pressure, mixing times, and fill volumes. The result? Higher batch consistency, better yields, and a product quality profile that outpaces what manual oversight alone could ever achieve. Accelerated R&D and Formulation Development. AI-powered molecular modeling and formulation simulation tools are compressing drug development timelines significantly. What once took months of bench-level trial and error can now be narrowed down to the most promising candidates in a fraction of the time. Regulatory Intelligence. Navigating the patchwork of global regulatory requirements — from the US FDA and EU EMA to emerging market health authorities — is a formidable challenge. AI tools that monitor regulatory changes, flag compliance gaps, and even draft submission-ready documentation are becoming standard infrastructure for global pharmaceutical manufacturing companies. At Kilitch Healthcare, our innovation-first DNA keeps us at the forefront of these advancements. With over 50 highly qualified scientists in our R&D and Quality Control division, we’ve embraced technology-driven approaches to ensure every product we manufacture meets the highest international standards — consistently and at scale. Rule #3: Specialization Beats Commoditization Every Time Here’s a hard truth for any global pharmaceutical manufacturing company playing in 2026: competing on cost alone is a race to the bottom. The manufacturers winning today have made a strategic pivot toward high-value, complex, and differentiated product categories. Sterile injectables and biologics are at the top of that list. The global market for sterile injectables continues to grow at an impressive rate, driven by the rise of oncology, immunology, and rare disease therapies. Manufacturing these products requires a level of technical sophistication, facility investment, and regulatory know-how that creates genuine barriers to entry — and genuine premium margins for those who do it right. Ophthalmic formulations represent another high-growth, high-complexity segment. The aging global population, combined with rising incidences of dry eye, glaucoma, and macular degeneration, is driving unprecedented demand for sophisticated ophthalmic products. Companies that have invested in advanced ophthalmic manufacturing capabilities are reaping the rewards. Prefilled Syringes (PFS) and Ready-to-Use formats are transforming the injectables market. Healthcare systems globally are demanding delivery formats that reduce preparation errors, improve dosing accuracy, and streamline clinical workflows. The pharmaceutical manufacturer that can produce high-quality PFS at scale for global markets holds a significant strategic advantage. This is precisely the territory where Kilitch
The Future of Pharmaceutical Manufacturing and Innovation in Global Healthcare

How Innovation is Transforming the Pharmaceutical Manufacturing Industry The pharmaceutical industry is constantly evolving to meet the changing needs of healthcare systems around the world. New diseases, growing populations, and increased demand for advanced treatments are driving innovation in medicine production. This is why a modern Pharmaceutical manufacturing company must continuously improve its technology, processes, and quality systems. Today, a reliable Pharmaceutical manufacturer in India uses advanced machinery, automation, and digital monitoring systems to produce medicines more efficiently. These technologies help ensure consistent quality while reducing production time. A professional Pharma manufacturing company focuses on innovation to deliver safe and effective medicines that meet global healthcare standards. Organizations like Kilitch Healthcare are investing in modern infrastructure and research to support future healthcare needs. Their commitment to innovation helps improve manufacturing efficiency and maintain high product quality. The Role of Automation and Technology in Modern Pharmaceutical Production Technology plays a major role in improving the safety and reliability of pharmaceutical manufacturing. Automated systems help reduce human error and ensure accurate measurement of ingredients. These systems also allow manufacturers to monitor production conditions in real time and quickly respond to potential issues. A trusted GMP certified pharmaceutical company uses advanced quality control equipment to test medicines before releasing them to the market. These tests confirm that products meet safety and performance standards. A professional WHO-GMP pharmaceutical manufacturer also uses digital record-keeping systems to track production data and maintain regulatory compliance. These systems help ensure transparency and accountability in manufacturing operations. Key Technologies Used in Modern Pharmaceutical Manufacturing Important Innovation Tools These technologies help maintain the high standards expected from a reliable Global pharmaceutical manufacturing company. The Growing Importance of Specialized Manufacturing in Healthcare Healthcare providers are increasingly relying on specialized medicines to treat complex medical conditions. This trend has increased demand for services such as Sterile injectable manufacturing, Aseptic pharmaceutical manufacturing, and Ophthalmic drug manufacturing. An experienced Injectable drug manufacturer produces medicines used in emergency care, surgeries, and vaccination programs. Because these medicines are delivered directly into the body, they must be produced under strict sterile conditions. A trusted Sterile injectable pharmaceutical company ensures that every product meets high safety standards. Similarly, a skilled Ophthalmic pharmaceutical manufacturer produces eye care medicines designed to treat infections, allergies, and vision-related conditions. A reliable Eye drops manufacturer follows strict hygiene procedures to maintain product safety. Organizations like Kilitch Healthcare continue to expand their specialized manufacturing capabilities to meet growing healthcare demand. The Role of Lyophilized and Sterile Ophthalmic Manufacturing A professional Lyophilized injectable manufacturer uses freeze-drying technology to improve product stability and extend shelf life. This method is especially useful for vaccines and biological medicines that require long-term storage. At the same time, Sterile ophthalmic manufacturing ensures that eye care medicines remain safe and effective. A trusted Ophthalmic formulations manufacturer uses advanced sterilization and packaging systems to maintain product quality. These specialized manufacturing processes help improve patient outcomes and support modern healthcare systems. Expanding Global Opportunities Through Pharmaceutical Export As healthcare demand continues to grow worldwide, pharmaceutical companies are expanding their presence in international markets. A reliable Pharmaceutical exporter from India plays a vital role in supplying medicines to countries that depend on imported healthcare products. An experienced Injectable exporter ensures that temperature-sensitive medicines are transported safely using controlled packaging and storage systems. Proper logistics management helps maintain product quality during shipping. A professional Pharma export company also manages regulatory documentation and compliance requirements for international trade. These efforts help ensure smooth delivery of medicines to healthcare providers across different regions. Companies like Kilitch Healthcare continue to strengthen their global network by delivering high-quality pharmaceutical products to international markets. Building a Sustainable Future for Global Healthcare Sustainability is becoming an important focus in pharmaceutical manufacturing. Companies are adopting environmentally friendly practices to reduce waste, conserve energy, and protect natural resources. These efforts help support long-term growth in the healthcare industry. A responsible Global pharmaceutical manufacturing company invests in energy-efficient equipment, safe waste management systems, and responsible sourcing of raw materials. These practices help reduce environmental impact while maintaining high production standards. Healthcare providers and patients are increasingly choosing manufacturers that demonstrate commitment to sustainability and ethical practices. This shift is encouraging pharmaceutical companies to adopt greener production methods and improve operational efficiency. Conclusion In conclusion, the future of the pharmaceutical industry depends on innovation, quality, and global collaboration. A modern Pharmaceutical manufacturing company must continuously invest in technology and skilled professionals to meet growing healthcare demands. Trusted organizations like Kilitch Healthcare play an important role in shaping the future of medicine production by maintaining high standards and expanding their global reach. Their commitment to quality and innovation supports healthcare providers and patients worldwide. As the pharmaceutical industry continues to evolve, working with a reliable Global pharmaceutical manufacturing company will remain essential for ensuring safe, effective, and accessible medicines for everyone.
The Role of Aseptic Pharmaceutical Manufacturing in Ensuring Safe Medicines

What is Aseptic Pharmaceutical Manufacturing and Why It Matters In the pharmaceutical industry, patient safety is always the top priority. Medicines must be produced in environments that prevent contamination and maintain product quality. This is where Aseptic pharmaceutical manufacturing becomes extremely important. It is a specialized process used to produce sterile medicines such as injections, vaccines, and eye care products. A reliable Pharmaceutical manufacturing company follows strict procedures to ensure that medicines are prepared in clean and controlled conditions. These procedures include maintaining sterile equipment, using purified raw materials, and monitoring production environments. A professional Pharmaceutical manufacturer in India invests in advanced technology to meet these safety requirements. Healthcare providers depend on trusted manufacturers to deliver safe medicines that protect patient health. Companies like Kilitch Healthcare focus on maintaining modern facilities and trained staff to ensure consistent product quality and reliable supply. How Aseptic Manufacturing Ensures Product Safety Aseptic production involves preparing medicines in sterile environments where the risk of contamination is minimized. This process is especially important for medicines that are injected directly into the body. Even a small amount of bacteria or dust can cause serious health problems for patients. A professional Sterile injectable pharmaceutical company uses clean rooms equipped with air filtration systems that remove harmful particles from the environment. Staff members wear protective clothing such as gloves, masks, and sterile gowns to maintain hygiene during production. An experienced Injectable drug manufacturer also performs regular equipment sterilization and quality testing to ensure product safety. These practices help maintain consistent quality in Sterile injectable manufacturing and protect patients from harmful infections. Key Components of Aseptic Manufacturing Essential Safety Measures These safety measures help maintain the high standards expected from a responsible Pharma manufacturing company. The Importance of GMP and WHO-GMP Certification in Aseptic Production Quality certification plays a major role in maintaining safety in pharmaceutical manufacturing. A GMP certified pharmaceutical company follows strict guidelines that control every step of the production process. These guidelines ensure that medicines are produced consistently and meet regulatory requirements. A WHO-GMP pharmaceutical manufacturer meets internationally recognized standards that allow medicines to be distributed in global markets. Certification demonstrates that the manufacturing facility follows proper hygiene practices, quality control procedures, and documentation systems. Healthcare providers and distributors prefer to work with certified manufacturers because they trust the quality and reliability of their products. A professional Global pharmaceutical manufacturing company maintains regular inspections and continuous improvement programs to meet changing industry standards. Organizations like Kilitch Healthcare maintain strict compliance with international quality standards to ensure safe and reliable production of sterile medicines. The Role of Aseptic Manufacturing in Ophthalmic and Injectable Products Aseptic manufacturing is essential for producing both injectable medicines and eye care products. These products require high levels of purity because they come into direct contact with sensitive parts of the body. In Ophthalmic drug manufacturing, sterile conditions ensure that eye drops and eye solutions remain safe for patient use. A trusted Ophthalmic pharmaceutical manufacturer follows strict sterilization procedures to prevent contamination during production. A reliable Eye drops manufacturer uses automated filling and sealing machines to maintain product safety and accuracy. Similarly, in injectable production, a professional Lyophilized injectable manufacturer follows aseptic processes to maintain product stability and effectiveness. Why Sterile Ophthalmic Manufacturing is Critical Sterile ophthalmic manufacturing ensures that eye care medicines are produced in safe environments. A skilled Ophthalmic formulations manufacturer uses advanced filtration systems and quality testing methods to maintain product purity. These processes help reduce the risk of infections and improve patient outcomes, especially for individuals undergoing eye surgery or treatment for eye diseases. Global Demand for Safe Medicines and Export Opportunities The demand for safe and high-quality medicines is increasing worldwide. Many countries rely on imported pharmaceutical products to support their healthcare systems. A reliable Pharmaceutical exporter from India plays an important role in meeting this demand. An experienced Injectable exporter ensures that sterile medicines are transported safely using proper packaging and temperature-controlled storage. These measures help maintain product quality during shipping. A professional Pharma export company also manages regulatory documentation and quality verification required for international trade. A trusted Global pharmaceutical manufacturing company focuses on building long-term partnerships with healthcare providers around the world. Companies like Kilitch Healthcare continue to expand their global presence by maintaining strict safety standards and delivering reliable pharmaceutical products. Conclusion In conclusion, Aseptic pharmaceutical manufacturing is essential for producing safe and effective medicines. It protects patients from contamination, ensures product quality, and supports reliable healthcare services. A trusted Pharmaceutical manufacturing company that follows strict quality standards can deliver consistent and safe medicines to healthcare providers. Organizations like Kilitch Healthcare continue to invest in advanced technology and skilled professionals to meet growing global demand. As healthcare systems continue to expand worldwide, the role of a reliable Global pharmaceutical manufacturing company in maintaining safe production practices will remain critical for protecting patient health.
How Lyophilized Injectable Manufacturing Improves Medicine Stability and Shelf Life
Understanding the Importance of Lyophilized Injectable Manufacturing In the pharmaceutical industry, medicine stability is extremely important. Many medicines lose their effectiveness if they are exposed to moisture, heat, or light for long periods. This is why Lyophilized injectable manufacturer services are becoming more important in modern healthcare. Lyophilization, also known as freeze-drying, is a process that removes water from medicines to improve their stability and shelf life. A reliable Pharmaceutical manufacturing company uses advanced technology to produce stable and safe medicines that can be stored for longer periods. Hospitals and healthcare providers depend on these medicines, especially in emergency situations where reliable treatment is required. A professional Pharmaceutical manufacturer in India focuses on maintaining strict quality standards to ensure that lyophilized medicines remain effective until they are used. Companies like Kilitch Healthcare invest in modern production systems to support the growing demand for stable injectable medicines. Their expertise in manufacturing helps healthcare providers deliver safe and effective treatments to patients. How the Lyophilization Process Works in Pharmaceutical Manufacturing The lyophilization process involves freezing the medicine and then removing moisture through a controlled drying process. This method helps preserve the chemical structure of the medicine and prevents bacterial growth. As a result, the medicine remains stable for a longer period without losing its effectiveness. A trusted Sterile injectable pharmaceutical company carefully monitors temperature, pressure, and humidity during the production process. These controls ensure that the final product meets strict quality and safety standards. Key Steps in the Lyophilization Process Important Production Stages These steps are essential for maintaining product safety and reliability in Sterile injectable manufacturing. A skilled Injectable drug manufacturer ensures that each batch is produced consistently and meets regulatory requirements. Organizations like Kilitch Healthcare follow strict production procedures to ensure that lyophilized medicines maintain high quality and performance. Benefits of Lyophilized Medicines in Healthcare Lyophilized medicines offer several advantages compared to traditional liquid medicines. One of the main benefits is improved shelf life. Because moisture is removed during production, the medicine can be stored for longer periods without losing its effectiveness. Another important benefit is easier transportation. Freeze-dried medicines are lighter and more stable, making them suitable for distribution to remote areas and international markets. A reliable Pharma manufacturing company ensures that these medicines remain safe during storage and transportation. Healthcare providers also prefer lyophilized medicines because they can be reconstituted quickly before use. This makes them ideal for emergency care and critical treatments. A professional GMP certified pharmaceutical company ensures that lyophilized products meet strict quality standards and remain safe for patient use. These standards help maintain consistency and reliability in medicine production. The Role of Aseptic Pharmaceutical Manufacturing in Lyophilized Products Because lyophilized medicines are often used in injections, they must be produced in sterile environments. This is where Aseptic pharmaceutical manufacturing plays a critical role. The process ensures that medicines are prepared without contamination from bacteria or other harmful microorganisms. A trusted WHO-GMP pharmaceutical manufacturer uses clean room technology, advanced filtration systems, and automated equipment to maintain sterile conditions. Staff members follow strict hygiene practices to protect product safety. Modern pharmaceutical facilities use continuous monitoring systems to detect any contamination risks during production. A reliable Global pharmaceutical manufacturing company invests in these technologies to maintain high safety standards and comply with international regulations. Companies like Kilitch Healthcare continue to strengthen their manufacturing processes by adopting advanced aseptic technologies. Their focus on quality helps build trust among healthcare providers and patients. Expanding Global Demand for Lyophilized Injectable Export The demand for stable and long-lasting medicines is increasing worldwide. Many countries rely on imported pharmaceutical products to meet healthcare needs. A reliable Pharmaceutical exporter from India plays a key role in delivering lyophilized medicines to international markets. An experienced Injectable exporter ensures that medicines are transported safely using proper packaging and temperature control systems. These measures help maintain product quality during long-distance shipping. A professional Pharma export company also manages regulatory approvals and documentation required for global distribution. This ensures smooth delivery of medicines to hospitals and healthcare providers in different countries. Organizations like Kilitch Healthcare continue to expand their international presence by providing high-quality lyophilized medicines that meet global healthcare standards. Conclusion In conclusion, the role of a Lyophilized injectable manufacturer is essential for improving medicine stability and ensuring reliable treatment. The lyophilization process helps protect medicines from moisture, extends shelf life, and supports safe transportation. A trusted Pharmaceutical manufacturing[KR1] company that follows strict quality standards can deliver safe and effective injectable products to healthcare providers. Companies like Kilitch Healthcare continue to invest in advanced technology and skilled professionals to meet growing global demand. As healthcare systems continue to expand worldwide, the importance of working with a reliable Global pharmaceutical manufacturing company will remain critical for ensuring safe and consistent medicine supply. [KR1]
The Importance of Ophthalmic Drug Manufacturing in Protecting Eye Health

Understanding the Role of Ophthalmic Drug Manufacturing in Modern Healthcare Eye health is an essential part of overall well-being. Millions of people around the world depend on eye care medicines to treat infections, allergies, dry eye conditions, and vision problems. Because the eyes are very sensitive organs, medicines used for eye treatment must be produced under extremely clean and controlled conditions. This is why Ophthalmic drug manufacturing plays a critical role in modern healthcare. A reliable Pharmaceutical manufacturing company ensures that eye care medicines are safe, effective, and free from contamination. Healthcare providers trust experienced manufacturers to produce high-quality ophthalmic products that protect patient safety. A professional Pharmaceutical manufacturer in India uses advanced technology and strict quality standards to meet growing demand for eye care medicines. Companies like Kilitch Healthcare focus on maintaining modern facilities and skilled teams to deliver safe ophthalmic solutions. Their commitment to quality helps support hospitals, clinics, and pharmacies in providing reliable treatment to patients. Why Sterile Ophthalmic Manufacturing Is Essential for Patient Safety Medicines used in eye treatment must be completely sterile because even a small amount of contamination can cause serious infections. This is why Sterile ophthalmic manufacturing is one of the most important processes in the pharmaceutical industry. Manufacturers must follow strict hygiene procedures and quality control systems to maintain product safety. A trusted Ophthalmic pharmaceutical manufacturer produces eye care medicines in controlled environments known as clean rooms. These facilities use advanced air filtration systems to remove dust and microorganisms. Staff members follow strict hygiene practices and wear protective clothing to prevent contamination during production. A reliable Eye drops manufacturer carefully monitors every stage of production, from raw material selection to final packaging. Each batch of medicine is tested to ensure safety, purity, and effectiveness before it reaches the market. Key Safety Measures in Ophthalmic Manufacturing Essential Quality Practices These practices help maintain the high standards expected from a professional Pharma manufacturing company. Types of Ophthalmic Formulations Produced by Modern Manufacturers Modern healthcare requires a wide range of eye care medicines to treat different conditions. A skilled Ophthalmic formulations manufacturer produces various products designed to meet specific patient needs. Common ophthalmic products include: Each product must be carefully formulated to ensure comfort, safety, and effectiveness. A professional GMP certified pharmaceutical company follows strict production guidelines to maintain consistent product quality. Organizations like Kilitch Healthcare continue to expand their ophthalmic product range to meet growing healthcare demand. Their focus on research and quality helps improve patient outcomes and support eye care professionals. The Role of Technology in Modern Ophthalmic Drug Manufacturing Technology plays a major role in improving the safety and efficiency of Ophthalmic drug manufacturing. Modern equipment allows manufacturers to produce medicines with greater precision and consistency. Automated systems help reduce human error and ensure accurate dosage in every product. A reliable WHO-GMP pharmaceutical manufacturer invests in advanced machinery, quality testing laboratories, and digital monitoring systems. These technologies help maintain strict production standards and improve product reliability. Another important technology used in ophthalmic production is automated filling and sealing equipment. This equipment ensures that eye care medicines are packaged safely and remain sterile until use. A professional Sterile injectable pharmaceutical company often uses similar technology to maintain product safety across different types of medicines. Companies like Kilitch Healthcare continuously upgrade their manufacturing facilities to adopt new technologies. Their commitment to innovation helps maintain high-quality standards and support long-term growth in the pharmaceutical industry. Expanding Global Access Through Ophthalmic Product Export The demand for eye care medicines is increasing worldwide due to aging populations, digital screen usage, and rising awareness of eye health. A reliable Pharmaceutical exporter from India plays an important role in delivering ophthalmic products to international markets. An experienced Pharma export company ensures that medicines are packaged securely and transported under controlled conditions. Proper storage and handling help maintain product quality during long-distance shipping. A trusted Global pharmaceutical manufacturing company also manages regulatory approvals and quality documentation required for international distribution. These efforts help build strong relationships with healthcare providers around the world. Organizations like Kilitch Healthcare continue to expand their global reach by delivering safe, high-quality ophthalmic products to multiple countries. Conclusion In conclusion, Ophthalmic drug manufacturing is essential for protecting eye health and improving patient quality of life. Because eye care medicines require high levels of purity and precision, manufacturers must follow strict safety and quality standards. A reliable Pharmaceutical manufacturing company ensures that ophthalmic products are safe, effective, and consistent. Companies like Kilitch Healthcare continue to invest in advanced technology, skilled professionals, and strong quality systems to meet growing global demand. As healthcare needs continue to grow, the role of a professional Global pharmaceutical manufacturing company in producing safe ophthalmic medicines will remain vital for supporting patient health worldwide.
The Growing Demand for Sterile Injectable Manufacturing in Modern Healthcare
Why Sterile Injectable Medicines Are Essential in Healthcare In modern healthcare, injectable medicines play a critical role in treating serious medical conditions. Hospitals and clinics rely on injections for emergency care, surgeries, vaccinations, and chronic disease management. Because these medicines enter directly into the bloodstream, they must be produced under extremely clean and controlled conditions. This is why Sterile injectable manufacturing has become one of the most important services provided by a reliable Pharmaceutical manufacturing company. A professional Pharmaceutical manufacturer in India understands the importance of producing safe and high-quality injectable medicines. These products must be free from contamination and manufactured according to strict safety standards. Healthcare providers depend on trusted manufacturers to deliver medicines that are safe, effective, and ready for immediate use. Companies like Kilitch Healthcare focus on maintaining advanced production facilities and strong quality systems to meet the growing demand for sterile injectable medicines. As healthcare services expand worldwide, the need for reliable injectable production continues to increase. Understanding the Process of Sterile Injectable Manufacturing The production of injectable medicines involves several carefully controlled steps to ensure product safety. A responsible Injectable drug manufacturer follows strict procedures during preparation, filling, sealing, and packaging. Each stage of production is monitored to maintain high quality standards. A trusted Sterile injectable pharmaceutical company uses specialized equipment such as clean rooms, sterile filtration systems, and automated filling machines. These technologies help reduce the risk of contamination and ensure consistent product quality. Key Steps in the Injectable Manufacturing Process Important Production Stages These steps are essential for maintaining the safety and reliability of injectable medicines produced by a professional Pharma manufacturing company. The Role of Aseptic Pharmaceutical Manufacturing in Patient Safety One of the most critical aspects of injectable production is Aseptic pharmaceutical manufacturing. This process ensures that medicines are prepared in a sterile environment without exposure to harmful microorganisms. Even small contamination can cause serious health risks for patients, especially those with weakened immune systems. A skilled WHO-GMP pharmaceutical manufacturer invests in advanced clean room technology and strict hygiene procedures to maintain sterile conditions. Staff members wear protective clothing, and air filtration systems continuously remove particles from the production area. Another important area of injectable production is freeze-dried medicines. A specialized Lyophilized injectable manufacturer uses a process called lyophilization to remove moisture from medicines. This method improves product stability and allows medicines to be stored for longer periods without losing effectiveness. Organizations like Kilitch Healthcare follow strict quality control procedures to ensure that every injectable product meets international safety standards. Their commitment to quality helps protect patient health and build trust with healthcare providers. The Importance of Quality Certification in Injectable Manufacturing Quality certification is essential for ensuring the safety and reliability of injectable medicines. A GMP certified pharmaceutical company follows strict guidelines that control every aspect of production, from raw material handling to final product testing. These standards help maintain consistent product quality and reduce the risk of errors. A WHO-GMP pharmaceutical manufacturer meets internationally recognized standards that allow medicines to be distributed in global markets. Certification also demonstrates that the company is committed to patient safety and regulatory compliance. Healthcare providers and distributors prefer to work with certified manufacturers because they trust the quality of their products. A reliable Global pharmaceutical manufacturing company maintains detailed records, conducts regular inspections, and continuously improves production processes to meet changing industry requirements. Expanding Global Opportunities Through Injectable Export The demand for injectable medicines is increasing worldwide, especially in developing countries where healthcare infrastructure is growing rapidly. A reliable Pharmaceutical exporter from India plays an important role in supplying essential medicines to international markets. An experienced Injectable exporter ensures that medicines are transported safely using temperature-controlled packaging and secure logistics systems. Proper handling during shipping helps maintain product quality and effectiveness. A trusted Pharma export company also manages documentation, regulatory approvals, and quality checks required for international distribution. These efforts help build strong partnerships with healthcare providers around the world. Companies like Kilitch Healthcare continue to expand their global presence by maintaining high manufacturing standards and reliable export services. Their focus on quality and innovation supports healthcare systems in many countries. Conclusion In conclusion, Sterile injectable manufacturing is a vital part of modern healthcare. Injectable medicines save lives in emergencies, support surgical procedures, and help manage chronic diseases. Because of their importance, these medicines must be produced with the highest level of safety and precision. A trusted Pharmaceutical manufacturing company that follows strict quality standards can ensure reliable production and consistent supply of injectable medicines. Organizations like Kilitch Healthcare continue to invest in advanced technology and skilled professionals to meet growing global demand. As healthcare systems expand worldwide, the role of a professional Global pharmaceutical manufacturing company in producing safe and effective injectable medicines will become even more important.
The Importance of GMP and WHO-GMP Certification in Pharmaceutical Manufacturing

What is GMP Certification in the Pharmaceutical Industry In the pharmaceutical industry, quality and safety are extremely important because medicines directly affect human health. That is why every responsible GMP certified pharmaceutical company follows strict rules known as Good Manufacturing Practices (GMP). These rules ensure that medicines are produced in clean, controlled environments and meet consistent quality standards. A professional Pharmaceutical manufacturing company must carefully manage every step of the production process. This includes selecting high-quality raw materials, maintaining equipment, training staff, and performing detailed quality checks. By following GMP guidelines, companies reduce the risk of contamination, product defects, and safety issues. Many healthcare providers prefer to work with a reliable Pharmaceutical manufacturer in India that follows GMP standards because it ensures safe and effective medicines. Organizations like Kilitch Healthcare maintain strict manufacturing procedures to protect patient health and build trust with healthcare partners. GMP certification is not just a legal requirement; it is also a sign of professionalism and responsibility. It shows that the company is committed to producing medicines that meet national and international safety standards. Understanding WHO-GMP Certification and Global Standards While GMP certification is important, WHO-GMP pharmaceutical manufacturer certification is even more valuable for companies that operate in international markets. The World Health Organization (WHO) sets global standards for pharmaceutical manufacturing to ensure consistent quality across different countries. A certified WHO-GMP pharmaceutical manufacturer must follow strict quality systems, maintain detailed documentation, and undergo regular inspections by regulatory authorities. These inspections verify that the manufacturing facility meets international safety and hygiene standards. A trusted Global pharmaceutical manufacturing company invests in modern technology and quality management systems to maintain WHO-GMP compliance. This certification helps companies export medicines to multiple countries and build strong relationships with global healthcare providers. Companies like Kilitch Healthcare continuously upgrade their manufacturing facilities to meet international quality requirements. By maintaining WHO-GMP certification, they demonstrate their commitment to patient safety and reliable medicine supply. Key Requirements for WHO-GMP Compliance To achieve and maintain WHO-GMP certification, pharmaceutical companies must follow several important requirements. Essential Compliance Standards These practices help ensure that medicines produced by a Pharma manufacturing company remain safe, effective, and consistent. How GMP Certification Supports Sterile Injectable Manufacturing The importance of GMP standards becomes even greater in specialized manufacturing processes such as Sterile injectable manufacturing. Injectable medicines are delivered directly into the body, so they must be completely free from contamination. An experienced Injectable drug manufacturer follows strict sterile procedures to ensure product safety. A reliable Sterile injectable pharmaceutical company uses advanced clean room technology, air filtration systems, and automated equipment to maintain a controlled production environment. Another important process in injectable production is Aseptic pharmaceutical manufacturing. This method involves producing medicines in sterile conditions to prevent contamination during the manufacturing process. It requires highly trained staff, specialized equipment, and continuous monitoring. A skilled Lyophilized injectable manufacturer also follows GMP guidelines when producing freeze-dried medicines. This technology improves product stability and allows medicines to be stored for longer periods without losing effectiveness. The Role of GMP in Ophthalmic Drug Manufacturing GMP standards are equally important in Ophthalmic drug manufacturing because eye care products require extremely high levels of purity. Even small contaminants can cause irritation or infection in the eyes. A professional Ophthalmic pharmaceutical manufacturer produces sterile eye care medicines such as eye drops, gels, and solutions. A reliable Eye drops manufacturer ensures that products are prepared in clean environments and tested thoroughly before distribution. Why Sterile Ophthalmic Manufacturing is Critical Sterile ophthalmic manufacturing ensures that eye care medicines remain safe for patient use. A trusted Ophthalmic formulations manufacturer follows strict sterilization procedures and quality control systems to maintain product safety. Modern manufacturing facilities use advanced filtration systems, automated filling machines, and controlled storage conditions to maintain sterile environments. Companies like Kilitch Healthcare invest in advanced technology to ensure consistent quality in ophthalmic products. The Importance of Certification for Pharmaceutical Export and Global Trust Certification plays a major role in international trade. Many countries only import medicines from certified manufacturers that meet global quality standards. A reliable Pharmaceutical exporter from India must maintain GMP and WHO-GMP certification to enter international markets. An experienced Injectable exporter ensures that medicines are produced, stored, and transported according to strict quality requirements. A trusted Pharma export company also maintains proper documentation and compliance with international regulations. A professional Global pharmaceutical manufacturing company builds long-term partnerships by delivering safe, high-quality medicines to healthcare providers worldwide. Certification helps companies demonstrate reliability, professionalism, and commitment to patient safety. Conclusion In conclusion, GMP and WHO-GMP certifications are essential for maintaining safety and quality in pharmaceutical production. A reliable Pharmaceutical manufacturing company that follows these standards can produce safe medicines, protect patient health, and build trust in global markets. Organizations like Kilitch Healthcare continue to invest in quality systems, modern technology, and skilled professionals to maintain high manufacturing standards. As healthcare demand continues to grow worldwide, working with a certified Global pharmaceutical manufacturing company will remain a key factor in ensuring safe and reliable medicine supply.
How a Pharmaceutical Manufacturing Company Supports Safe and Reliable Healthcare

Understanding the Role of a Pharmaceutical Manufacturing Company in Modern Medicine A Pharmaceutical manufacturing company plays a very important role in the healthcare system. Every hospital, clinic, and pharmacy depends on reliable medicine supply to treat patients safely. Without proper manufacturing, doctors would not be able to provide the right treatment at the right time. Today, healthcare needs are increasing around the world. New diseases, aging populations, and growing demand for better treatment require strong pharmaceutical production systems. A trusted Pharmaceutical manufacturer in India focuses on quality, safety, and efficiency to meet these healthcare demands. Companies like Kilitch Healthcare work hard to maintain strict quality standards in medicine production. By following international guidelines and using advanced technology, they ensure that medicines are safe for patients. A professional Pharma manufacturing company not only produces medicines but also supports healthcare providers by maintaining a steady supply of essential products. Why Quality Standards Are Important in Pharmaceutical Manufacturing Quality is the foundation of the pharmaceutical industry. A GMP certified pharmaceutical company follows strict manufacturing practices to ensure that medicines are safe, effective, and consistent. These standards cover every stage of production, including raw material selection, equipment maintenance, and product testing. A WHO-GMP pharmaceutical manufacturer follows international quality guidelines recognized by global health authorities. This certification shows that the company is committed to producing medicines that meet global safety standards. Key Quality Practices Followed by a Pharmaceutical Manufacturing Company Essential Quality Measures A reliable Global pharmaceutical manufacturing company follows these practices to maintain consistent quality and build trust among healthcare providers. The Importance of Sterile Injectable Manufacturing in Healthcare Modern healthcare relies heavily on injectable medicines. Treatments for infections, chronic diseases, and emergency conditions often require injections. That is why Sterile injectable manufacturing is one of the most important processes in the pharmaceutical industry. An experienced Injectable drug manufacturer uses advanced equipment and controlled environments to produce sterile medicines. A trusted Sterile injectable pharmaceutical company must maintain strict hygiene standards to protect patient safety. How Aseptic Pharmaceutical Manufacturing Ensures Patient Safety Aseptic pharmaceutical manufacturing involves producing medicines in a sterile environment where contamination risks are minimized. This process uses clean rooms, air filtration systems, and protective clothing to maintain product safety. Companies like Kilitch Healthcare invest in advanced technology to ensure safe production and consistent supply of injectable medicines. The Role of Pharmaceutical Exporters in Global Healthcare Medicine production is not limited to local markets. Many countries rely on imported medicines to meet healthcare needs. A reliable Pharmaceutical exporter from India plays a key role in delivering high-quality medicines to international markets. An experienced Injectable exporter ensures that medicines are transported safely using temperature-controlled packaging and secure shipping methods. Why Global Partnerships Matter in Pharmaceutical Manufacturing A trusted Pharma export company builds strong relationships with distributors and hospitals worldwide. A responsible Global pharmaceutical manufacturing company focuses on long-term collaboration, quality assurance, and reliable delivery systems. Conclusion In conclusion, a reliable Pharmaceutical manufacturing company is essential for safe and effective healthcare. By following strict quality standards and maintaining strong supply chains, companies like Kilitch Healthcare continue to support patient health and global medicine access
Why Pharmaceutical Contract Manufacturing Is a Smart Strategy for Growing Pharma Businesses

Understanding Pharmaceutical Contract Manufacturing Many pharmaceutical companies aim to expand their product range and enter new markets, but not all of them have the infrastructure or resources to build their own manufacturing facilities. In such situations, Pharmaceutical contract manufacturing becomes a practical and cost-effective solution. This business model allows companies to outsource production to an experienced Pharmaceutical manufacturing company while focusing on branding, sales, and market expansion. Working with a professional Pharma manufacturing company helps businesses reduce operational risks and improve efficiency. Instead of investing large amounts of money in machinery, factory space, and skilled staff, companies can partner with a trusted manufacturer that already has the required facilities and expertise. Organizations like Kilitch Healthcare provide reliable contract manufacturing services supported by advanced technology, strict quality systems, and experienced professionals. By choosing an established manufacturing partner, pharmaceutical businesses can ensure consistent product quality and faster time-to-market. Another advantage of Pharmaceutical contract manufacturing is scalability. As market demand increases, manufacturers can quickly adjust production volumes without requiring additional investment from the client. This flexibility helps businesses respond to market changes and maintain a stable supply of medicines. Benefits of Working with a GMP Certified Pharmaceutical Company Quality and safety are essential in the pharmaceutical industry because medicines directly affect patient health. A GMP certified pharmaceutical company follows strict manufacturing guidelines to ensure that products are produced in clean, controlled environments. These guidelines cover every stage of production, including raw material selection, equipment maintenance, quality testing, and packaging. A WHO-GMP pharmaceutical manufacturer goes one step further by meeting internationally recognized quality standards. This certification demonstrates that the company is capable of producing medicines suitable for global distribution. Many international buyers prefer to work with certified manufacturers because they trust the quality and reliability of their products. Partnering with a certified Pharmaceutical manufacturer in India also helps companies comply with regulatory requirements in different countries. This is especially important for businesses planning to export medicines to international markets. Major Advantages of Pharmaceutical Contract Manufacturing Contract manufacturing offers several operational and financial benefits for pharmaceutical companies of all sizes. It allows businesses to focus on product development and marketing while experienced professionals handle production activities. Key Business Benefits These advantages make Pharmaceutical contract manufacturing a preferred strategy for companies looking to grow in competitive pharmaceutical markets. The Growing Importance of Specialized Manufacturing Healthcare systems around the world are increasingly relying on specialized medicines such as injectable drugs and eye care products. This trend has created strong demand for Sterile injectable manufacturing and Ophthalmic drug manufacturing services worldwide. An experienced Injectable drug manufacturer uses advanced equipment and strict quality control procedures to produce safe injectable medicines. These products are commonly used in hospitals for emergency treatments, vaccinations, and surgical procedures. A reliable Sterile injectable pharmaceutical company ensures that every product is free from contamination and meets strict safety standards. Another important area of specialized manufacturing is lyophilized products. A skilled Lyophilized injectable manufacturer uses freeze-drying technology to remove moisture from medicines, improving their stability and shelf life. This process is particularly useful for vaccines and biological medicines that require long-term storage. Similarly, an experienced Ophthalmic pharmaceutical manufacturer produces medicines designed specifically for eye care. These products include eye drops, ointments, and sterile solutions used to treat infections, allergies, and other eye conditions. A reliable Eye drops manufacturer must follow strict hygiene and sterilization procedures to protect patient safety. Role of Sterile Ophthalmic Manufacturing in Eye Care Sterile ophthalmic manufacturing plays a critical role in protecting eye health. Because the eyes are highly sensitive, even small amounts of contamination can cause serious infections or irritation. That is why a trusted Ophthalmic formulations manufacturer follows strict quality control processes during production. Modern manufacturing facilities use clean rooms, air filtration systems, and automated equipment to maintain sterile conditions. Companies like Kilitch Healthcare invest in advanced technologies to ensure that ophthalmic medicines meet international safety standards and deliver reliable results for patients. The Role of Export and Global Partnerships in Pharmaceutical Growth As healthcare demand continues to grow worldwide, pharmaceutical companies are expanding their reach to international markets. A reliable Pharmaceutical exporter from India plays an important role in delivering high-quality medicines to countries that depend on imported healthcare products. An experienced Injectable exporter ensures that temperature-sensitive medicines are transported safely using controlled packaging and storage systems. Proper logistics and documentation help maintain product quality during long-distance shipping. Working with a trusted Pharma export company also helps businesses build strong relationships with global distributors and healthcare providers. A professional Global pharmaceutical manufacturing company focuses on consistent quality, regulatory compliance, and timely delivery to support international healthcare systems. Companies like Kilitch Healthcare continue to strengthen their global presence by maintaining high manufacturing standards and reliable export services. Conclusion In today’s competitive pharmaceutical industry, Pharmaceutical contract manufacturing offers a smart and practical solution for business growth. It helps companies reduce costs, improve efficiency, and expand their product portfolio without investing heavily in infrastructure. By partnering with a trusted Pharmaceutical manufacturing company, businesses can access advanced technology, skilled professionals, and reliable production systems. Organizations like Kilitch Healthcare play a key role in supporting pharmaceutical companies with high-quality manufacturing and export services. As global demand for safe and effective medicines continues to rise, working with a reliable Global pharmaceutical manufacturing company will remain an essential strategy for long-term success in the healthcare industry.
The Rise of Preservative-Free Eye Drops: Revolutionizing Modern Eye Care
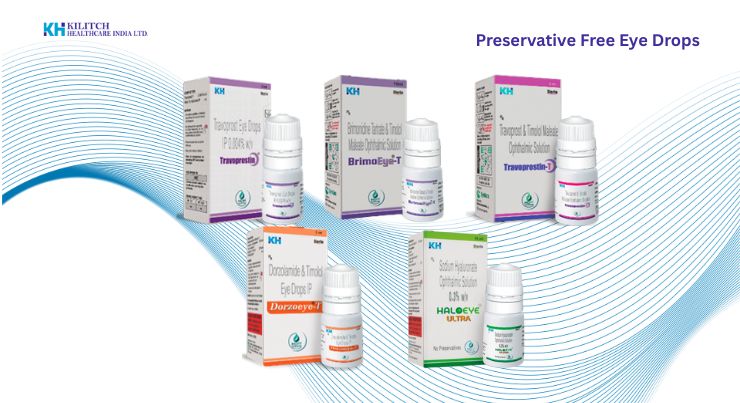
For decades, traditional eye drops have relied on chemical preservatives, such as Benzalkonium Chloride (BAK), to prevent bacterial contamination inside the bottle. However, as the global demand for long-term ophthalmic treatments—such as those for chronic dry eye syndrome and glaucoma—has surged, the medical community has recognized a significant drawback: prolonged exposure to preservatives can damage the ocular surface. Today, the pharmaceutical industry is undergoing a massive shift toward preservative-free eye drops. At Kilitch Healthcare, we are at the forefront of this transformation, leveraging cutting-edge, sterile dispensing technologies to ensure microbiological safety without compromising the patient’s long-term eye health. Why Are Preservative-Free Eye Drops Necessary? While preservatives effectively keep bacteria out of multi-use bottles, their prolonged use can lead to several adverse ocular effects: Eliminating preservatives ensures a highly tolerable and efficacious treatment, specifically for patients requiring daily, lifelong administration of ocular medication. The Challenge of Multidose Preservative-Free (MDPF) Systems Historically, preservative-free eye drops were only available in single-dose vials. While effective, single-dose units present their own challenges: they are often difficult for elderly patients to handle, generate significant plastic waste, and are generally more expensive to manufacture and purchase. The solution lies in advanced Multidose Preservative-Free (MDPF) systems. These sophisticated packaging technologies allow a sterile formulation to be dispensed from a standard-sized bottle over a prolonged period without the risk of bacterial ingress. Pioneering Ophthalmic Technologies at Kilitch Healthcare As a leading ophthalmic drug manufacturer, Kilitch Healthcare’s Eyekare Division is equipped to manufacture and package formulations using some of the world’s most sophisticated delivery systems. By utilizing state-of-the-art aseptic processing, we bring highly reliable MDPF solutions to the global market. 1. Advanced Multi-Dose Closing Tip Systems We utilize breakthrough multi-dose packaging that features specialized one-way valve systems. 2. Precision Metered-Dose Systems with Anti-Microbial Protection For highly sensitive formulations, we employ highly functional, precision-based dispensing mechanisms. The Future of Ophthalmic Manufacturing The transition toward preservative-free formulations is no longer just a trend; it is becoming a regulatory and clinical standard. Creating these complex systems requires immense expertise in pharmaceutical engineering, pharmacokinetics, and strict aseptic processing. From specialized homogenization to isolated sterile filling environments, the manufacturing process for MDPF systems is highly demanding. Partnering with a manufacturer equipped with international accreditations and World-Class GMP facilities is crucial to bringing these innovative eye care solutions to market safely, efficiently, and consistently. Conclusion As eye care becomes more sophisticated, patient comfort and safety remain the top priorities. Preservative-free eye drops delivered through intelligent multi-dose systems represent the perfect intersection of clinical efficacy and patient convenience. At Kilitch Healthcare, our commitment to innovation ensures that we continue to provide unparalleled ophthalmic manufacturing solutions to healthcare providers worldwide. Are you looking for an innovative, highly capable manufacturing partner for your ophthalmic product line? Contact Kilitch Healthcare today to learn more about our contract manufacturing services and proprietary eye care technologies.
