Inside Sterile Ophthalmic Manufacturing: Why Precision, Purity & Innovation Are Non-Negotiable

Inside Sterile Ophthalmic Manufacturing: Why Precision, Purity & Innovation Are Non-Negotiable Introduction: When a Drop is More Than a Drop The human eye is one of the most complex and sensitive organs in the body. It has no immune barrier equivalent to the skin, making it acutely vulnerable to contamination. A single particle — invisible to the naked eye — in an ophthalmic product can cause infection, inflammation, or permanent vision loss. This is why sterile ophthalmic manufacturing is not merely a regulatory requirement; it is the very foundation of safe, effective eye care. Across the globe, the ophthalmic drug market is growing rapidly — driven by an aging population, increasing prevalence of dry eye disease, glaucoma, and diabetic retinopathy, and a rising demand for surgical eye care products. Yet, this growth brings with it an equally rising responsibility: every eye drop, gel, injectable, or prefilled syringe that reaches a patient must be produced under conditions of absolute purity. At Kilitch Healthcare, sterile ophthalmic manufacturing is at the heart of what we do. Through our dedicated Eyekare Division, we have spent decades refining our processes, integrating internationally acclaimed technologies, and building a manufacturing ecosystem that sets benchmarks for quality, precision, and innovation. We take you inside the world of sterile ophthalmic manufacturing — what it means, what it demands, and what it takes to do it right. What Is Sterile Ophthalmic Manufacturing? Sterile ophthalmic manufacturing refers to the production of eye care formulations — including eye drops (solutions and suspensions), ophthalmic gels, ointments, intravitreal injectables, and prefilled syringes (PFS) — under strictly controlled conditions that eliminate microbial, particulate, and chemical contamination. Unlike oral drug manufacturing, ophthalmic products are applied directly to or injected into the eye. This means: Common product categories in sterile ophthalmic manufacturing include: topical eye drops and solutions, ophthalmic suspensions (where the active drug is in particle form), gel-forming eye drops for extended retention, intravitreal injectables for retinal disease, and prefilled syringes for surgical use. The Regulatory Backbone — Standards That Cannot Be Compromised Sterile ophthalmic manufacturing is one of the most heavily regulated segments of pharmaceutical production. Manufacturers must comply with a complex web of international and national standards — not just to gain market access, but to genuinely protect patient safety. Key Global Regulatory Frameworks Standard / Body Key Requirements for Ophthalmic Manufacturing WHO-GMP Aseptic processing, validated sterilization, in-process controls, QA documentation US FDA (21 CFR Part 211) Current Good Manufacturing Practices (cGMP) for finished pharmaceuticals, container/closure integrity EU GMP (Annex 1) Detailed requirements for sterile manufacturing — cleanroom classification, HVAC, environmental monitoring Schedule M (India) Revised Indian GMP standards now aligned with WHO-GMP for sterile products ISO 14644 Cleanroom and controlled environment standards for air classification Kilitch Healthcare’s manufacturing facility holds multiple international accreditations, reflecting our commitment to meeting the highest global standards — not just for domestic markets, but for regulated export markets across Europe, Africa, Asia, and beyond. The Manufacturing Process — Step by Step Producing a sterile ophthalmic product is a multi-stage, highly controlled process. Every step is governed by Standard Operating Procedures (SOPs), validated methods, and continuous environmental monitoring. Here is a breakdown of the key stages: Step 1: Formulation Development The journey begins in the laboratory, where formulation scientists design the product to meet precise physiological and pharmacological requirements. This includes balancing pH (ideally between 6.5–7.4, matching tear fluid), optimizing osmolarity (isotonic at approximately 300 mOsm/kg), selecting appropriate viscosity enhancers for retention, choosing between preserved and preservative-free formulations, and ensuring API stability and bioavailability at the ocular surface. Step 2: Cleanroom Environment Setup All sterile ophthalmic manufacturing takes place in classified cleanrooms — environments where airborne particles, temperature, humidity, and pressure differentials are rigorously controlled. The ISO 5 / Grade A environment (the strictest classification) is used for the actual filling operation. ISO 7 / Grade C areas are used for preparation and compounding. HVAC systems maintain laminar airflow, and continuous environmental monitoring tracks viable and non-viable particles. Step 3: Sterilization Sterilization is the most critical step in ophthalmic manufacturing. Two primary methods are used. Terminal Sterilization involves sterilizing the product after filling — typically by moist heat (autoclaving) or filtration + gamma irradiation. It provides the highest sterility assurance level. Aseptic Processing is used when the product cannot withstand heat sterilization. The drug solution is sterile-filtered (0.22 micron), and all components are sterilized separately before being assembled in the cleanroom. This method demands the highest level of operator training and environmental control. Step 4: Filling & Sealing Automated filling lines precisely dose the sterile formulation into pre-sterilized containers — multi-dose bottles, unit-dose vials, ampoules, or prefilled syringes. Filling is performed under Grade A laminar airflow conditions. Automated vision inspection systems check each container for fill volume accuracy, particulate contamination, and container integrity before sealing. Step 5: Quality Control & Batch Release No batch leaves the facility without passing a comprehensive quality control protocol. Key tests include sterility testing (per pharmacopoeial standards), pH and osmolarity verification, sub-visible and visible particulate matter analysis, container closure integrity testing (CCIT), preservative efficacy testing (for multi-dose products), microbial endotoxin / pyrogen testing, and stability studies to confirm shelf-life under real and accelerated conditions. Kilitch Healthcare’s Edge — Where Technology Meets Vision Kilitch Healthcare doesn’t just manufacture ophthalmic products — we engineer solutions that address unmet clinical needs. Through our Eyekare Division and our Innovations pipeline, we have integrated some of the world’s most advanced ophthalmic technologies into our manufacturing capabilities. ► Novelia Pure Flow Technology A pioneering French technology, introduced in India for the first time by Eyekare in 2014, the Novelia system is a multi-dose closing tip that prevents bacterial contamination without the need for preservatives. It has received approval for prescription and OTC ophthalmic products in markets worldwide. For patients with preservative sensitivity — a growing clinical concern — this technology is transformative. ► 3K Triple Protection System The world’s only patented metered-dose preservative-free delivery system, originally developed in Germany, the 3K system is designed for the precise, contamination-free delivery
Sterile Injectable Manufacturing | What Sets World-Class Facilities Apart

Sterile Injectable Manufacturing: Standards, Challenges, and What Sets World-Class Facilities Apart When doctors inject medication directly into a vein, muscle, or eye, the body has no natural filter. There is no digestive system to neutralize contamination. No barrier slows down a harmful particle. Therefore, everything that enters must be exactly right — in composition, concentration, and sterility. That is why sterile injectable manufacturing ranks among the most demanding disciplines in pharma. It combines precision engineering, rigorous science, and strict regulatory compliance. Together, these elements produce drug products that patients and clinicians trust completely. At Kilitch Healthcare, this is not just a service. It is a core commitment, backed by an internationally accredited facility and a culture of continuous innovation. What Is Sterile Injectable Manufacturing? Sterile injectable manufacturing involves producing drug products that enter the body through injection or infusion. These products bypass the body’s natural protective barriers. As a result, they must be completely free of microbial contamination, particulate matter, pyrogens, and foreign substances. The category includes several product types. Each one carries its own manufacturing complexity. Liquid Injectables are ready-to-use solutions filled into vials, ampoules, or prefilled syringes. Manufacturers must achieve precise formulation, pH control, and careful sterilization. These steps maintain chemical stability and biological safety. Lyophilized (Powder) Injectables use freeze-drying to remove moisture from a liquid drug product. This process extends shelf life and improves stability for sensitive molecules such as proteins, peptides, and biologics. Furthermore, manufacturers must develop and validate each lyophilization cycle for the specific formulation. Prefilled Syringes (PFS) deliver pre-measured doses in ready-to-use syringe barrels. They reduce dosing errors and limit contamination at the point of care. However, they also demand high-precision filling equipment and rigorous container compatibility testing. Additionally, sterile injectables serve critical therapeutic areas. These include oncology, ophthalmology, critical care, and immunology. In all these fields, manufacturing quality directly affects patient safety. The Regulatory Framework: Standards That Cannot Be Compromised Sterile injectable manufacturing operates under some of the strictest regulatory oversight in the world. To serve global markets, a manufacturer must comply with multiple international standards. There is no shortcut. WHO-GMP forms the global foundation. It covers facility design, personnel hygiene, process validation, and quality control systems. Moreover, WHO-GMP compliance is a prerequisite for supplying regulated markets across Asia, Africa, and beyond. US FDA 21 CFR Part 211 governs access to the American market — one of the most scrutinized regulatory environments globally. Manufacturers must maintain rigorous documentation, validated processes, and robust change control systems. EU GMP Annex 1 has recently undergone significant revision. It places heightened focus on Contamination Control Strategy (CCS) and barrier technologies such as isolators and RABS. In addition, it mandates comprehensive environmental monitoring throughout production. Schedule M (Revised) sets pharmaceutical manufacturing standards within India. It increasingly aligns domestic requirements with global benchmarks. ISO 14644 defines cleanroom classification — from Grade A through Grade D. Each grade corresponds to a specific stage of sterile manufacturing. Consequently, manufacturers must maintain constant monitoring, validated HVAC systems, and strict personnel discipline. Kilitch Healthcare’s facility spans 3,50,000 square feet in Mumbai. The team designed and built it to comply with all major international regulatory standards. As a result, it enables supply to both domestic and global regulated markets from a single, accredited location. The Sterile Injectable Manufacturing Process: From Raw Material to Release Understanding each step inside a GMP-compliant facility shows why quality cannot be compromised at any stage. Step 1 – Raw Material Testing and Release First, every ingredient enters a rigorous testing protocol. The Quality Assurance team checks the active pharmaceutical ingredient (API), excipients, and container components. They verify identity, purity, and quality before approving any raw material for production. Step 2 – Formulation Development Next, scientists develop and optimize the drug formulation. They evaluate stability, solubility, pH, osmolality, and container compatibility. For complex molecules, this phase also includes stress testing and accelerated stability studies. Step 3 – Cleanroom Preparation and Environmental Monitoring Before filling begins, the team certifies cleanrooms and disinfects all surfaces. They then initiate environmental monitoring. This measures airborne particle counts, microbial levels, and temperature. Only after the environment meets specification does production start. Step 4 – Filling Operations The team fills liquid injectables under aseptic conditions in Grade A zones (ISO Class 5). Grade B background environments surround these zones at all times. For powder injectables, operators choose between aseptic filling and terminal sterilization. Meanwhile, automated, high-precision lines fill prefilled syringes with minimal human contact. Step 5 – Sterilization The formulation determines the choice of sterilization method. Terminal sterilization (autoclaving) works well when the drug and container can tolerate the heat. However, for heat-sensitive molecules, manufacturers use aseptic processing instead. They sterilize all components separately and then combine them in a sterile environment. In either case, each method must achieve a Sterility Assurance Level (SAL) of 10⁻⁶ or better. Step 6 – Inspection and Leak Testing After filling, every unit goes through 100% visual inspection. Inspectors check for particulate matter, clarity, fill volume, and container integrity. Automated inspection systems work alongside trained human inspectors. Together, they provide multiple layers of assurance. Additionally, Container Closure Integrity Testing (CCIT) confirms hermetic seals throughout the product’s shelf life. Step 7 – Labeling, Packaging, and Batch Release Finally, the team labels and packages finished products. They then conduct Quality Control (QC) testing for sterility, endotoxin, potency, and container closure. A Qualified Person reviews all results before authorizing the batch for release. Core Challenges in Sterile Injectable Manufacturing Even experienced manufacturers face ongoing technical and operational challenges. These require continuous investment and vigilance. Maintaining Aseptic Conditions is the most fundamental challenge. A single breach in gowning protocol can compromise an entire batch. Similarly, a momentary lapse in airflow or an unvalidated equipment surface poses the same risk. The stakes are always high. Particulate Contamination Control requires constant environmental monitoring. Any out-of-trend result triggers a thorough investigation. Sources of contamination — including personnel, equipment wear, and packaging materials — each demand rigorous control. Lyophilization Cycle Development is scientifically complex. Developers must carefully
How Hydropure Technology is Shaping the Future of Pharma Packaging

In the critical fields of sterile injectable manufacturing and ophthalmic drug manufacturing, maintaining an absolute sterile environment isn’t just a regulatory requirement—it is a matter of patient safety. While the formulations inside the vials or ampoules are rigorously sterilized, the exterior packaging can often pose a hidden contamination risk when introduced into operating rooms or cleanroom environments. To combat this, the industry is turning to advanced sterilization techniques. At Kilitch Healthcare, a leading pharmaceutical manufacturer in India, we are pioneering the use of VH2O2 (Vapour Hydrogen Peroxide) Blister Technology to eliminate these risks and set a new benchmark for packaging safety. The Hidden Risk in Traditional Packaging Traditionally, terminally sterilized products are placed into standard blister packs. While the medication itself is safe, the exterior of the immediate product container (like the outside of a syringe or vial) can inadvertently harbor microscopic contaminants from the packaging process. When a surgeon or healthcare professional opens this standard blister pack in a sterile surgical field, that exterior contamination can be transferred, posing a severe risk of healthcare-associated infections (HAIs). For a global pharmaceutical manufacturing company, mitigating this risk is a top priority. What is Hydropure technology? Hydropure technology is a breakthrough innovation in aseptic pharmaceutical manufacturing. It utilizes Vapour Hydrogen Peroxide—a highly effective, low-temperature sterilizing agent—to completely sterilize the interior of the blister pack and the exterior of the immediate product contact surface just before the blister is sealed. Kilitch Healthcare is proud to be the 1st in India to adopt VH2O2 sterilized blister packs. This ensures that when the secondary packaging is peeled open in a clinical setting, both the drug and the exterior of its primary container are 100% sterile and intact. Key Benefits of VH2O2 Sterilization in Pharma By integrating this advanced technology, we provide an unparalleled level of safety for healthcare providers and patients alike: Elevating Global Contract Manufacturing Standards As a premier WHO-GMP pharmaceutical manufacturer, maintaining international manufacturing standards is at our core. The implementation of deeply validated, newly developed technologies like VH2O2 sterilization places Kilitch Healthcare among the elite ranks of manufacturers globally, rivaling the few companies in Europe that currently utilize this process. Whether you are looking for a partner for pharmaceutical contract manufacturing or require a reliable pharmaceutical exporter from India to supply highly regulated international markets, our world-class facility—spread across 350,000 Sq. Ft.—is equipped to meet your needs. We deploy Industry 4.0 GAMP standards to ensure every batch meets the most stringent global quality parameters. Conclusion The future of sterile injectable pharmaceutical companies depends on continuous innovation in both formulation and packaging. Hydropure technology is a testament to how advanced engineering can solve complex clinical challenges, ensuring that life-saving medications arrive safely from the factory floor to the patient’s bedside. Are you seeking a reliable, innovation-driven partner for your sterile manufacturing needs? Contact Kilitch Healthcare today to learn how our advanced capabilities and strict GMP compliance can elevate your product line.
Precision in Sterile Injectable Manufacturing: Quality, Compliance, and Efficiency at Every Milliliter
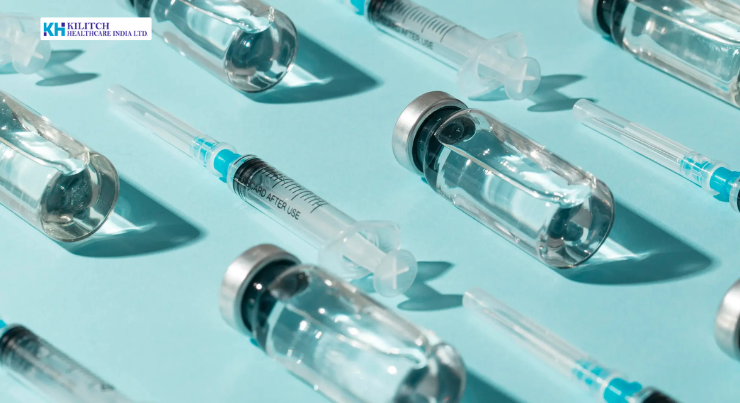
In today’s highly regulated healthcare landscape, the margin for error in pharmaceutical manufacturing is vanishingly small especially for sterile dosage forms. Sterile injectables remain a cornerstone of modern therapy in hospitals, critical care, and specialized treatment protocols. Their clinical impact is profound, but so are the technical and regulatory demands behind their production. For pharmaceutical companies, quality leaders, and contract partners, the challenge is clear: how to deliver consistent, contamination-free, and regulation-compliant products at scale without compromising efficiency or speed to market. As the global supply of injectable medicines grows and the pharmaceutical manufacturers in India expand their footprint, precision in sterile operations has become both a competitive differentiator and a compliance imperative. This article examines how precision, quality systems, and process efficiency converge in sterile injectable manufacturing and what decision-makers should evaluate when selecting a manufacturing partner. Precision and Quality Challenges in Sterile Pharmaceutical Manufacturing Sterile dosage forms are uniquely sensitive to process variability. Unlike non-sterile products, injectables are administered directly into the body, leaving no tolerance for microbial contamination, particulate matter, or dosage inconsistency. Achieving uniformity across every unit is therefore not just a technical objective; it is a patient safety mandate. Microbiological Control and Contamination Risk The primary challenge in producing sterile injectables is maintaining aseptic conditions throughout the manufacturing chain. Even minute lapses in environmental control, material handling, or operator procedures can compromise batch integrity. Common risk areas include: Precision in sterile operations requires layered controls engineering, procedural, and human to ensure that contamination risks are identified and mitigated at every step. Dosage Accuracy and Product Uniformity In injectable manufacturing, volumetric accuracy directly influences therapeutic outcomes. Variations at the microliter level can affect drug potency, stability, and safety. This places extraordinary demands on filling equipment calibration, in-process controls, and inspection systems. For pharmaceutical companies operating in regulated markets, failure to meet dosage specifications can result in batch rejections, regulatory observations, or product recalls making precision an operational and reputational priority. The Technical Process: From Aseptic Preparation to Final Inspection Achieving consistent quality in sterile manufacturing depends on a tightly controlled sequence of validated processes. While specific technologies vary by facility, best-in-class operations follow a structured approach to minimize risk and maximize repeatability. Aseptic Preparation and Component Sterilization The manufacturing cycle begins with the preparation of the drug solution and the sterilization of all contact components, including containers, closures, and transfer lines. Filtration systems, sterilization cycles, and material staging areas are designed to maintain product integrity before filling begins. At this stage, adherence to GMP good manufacturing practices ensures that materials, equipment, and documentation meet defined quality standards prior to entering controlled environments. Precision Filling and Sealing Operations Filling is the most critical step in sterile injectable manufacturing. Automated systems are used to dispense precise volumes into ampoules, vials, or other primary containers under aseptic conditions. Key process controls include: These systems are designed to deliver consistent dosage across high-volume production while maintaining sterility. Visual Inspection and Defect Detection Post-filling, each unit undergoes inspection to identify defects such as particulate matter, container cracks, or improper seals. Advanced inspection systems often combining automated and manual verification are employed to ensure that only compliant products proceed to packaging and release. This inspection phase is essential not only for quality assurance but also for regulatory defensibility, particularly in export-oriented operations. Regulatory and Quality Standards: Compliance as a Core Operating Principle In sterile manufacturing, regulatory compliance is inseparable from operational excellence. Manufacturing partners must demonstrate adherence to national and international guidelines, supported by robust quality management systems. Implementing GMP Across the Manufacturing Lifecycle GMP good manufacturing practices govern every aspect of sterile production from facility design and equipment qualification to documentation, training, and batch release. Core elements include: For pharmaceutical companies engaged in pharmaceutical contract manufacturing, these systems provide assurance that outsourced production meets regulatory expectations across markets. Quality Control, Documentation, and Audit Readiness Beyond physical controls, compliance depends on comprehensive documentation and traceability. Batch records, validation reports, stability data, and audit trails form the backbone of regulatory interactions. Facilities that integrate quality into daily operations rather than treating it as a checkpoint are better positioned to withstand inspections and sustain long-term partnerships. For global pharmaceutical brands, this audit readiness is a non-negotiable requirement when evaluating manufacturing partners in India or abroad. Efficiency, Scalability, and Business Impact While precision and compliance are foundational, modern pharmaceutical operations must also deliver efficiency and scalability. The ability to meet growing demand without compromising quality is critical for both market competitiveness and supply chain reliability. Automation and Throughput Optimization Automation reduces human intervention in critical zones, lowering contamination risk while increasing throughput. Integrated filling, inspection, and packaging lines enable higher batch volumes with consistent output supporting rapid scale-up for product launches or institutional supply contracts. For pharmaceutical companies navigating variable market demand, these efficiencies translate into shorter lead times, reduced waste, and improved cost control. Supply Chain Reliability for Institutional and Export Markets Hospital procurement teams and healthcare distributors require predictable supply and batch-to-batch consistency. Manufacturing operations that emphasize process standardization and capacity planning provide the reliability needed for institutional tenders and international distribution. In this context, India’s growing role in global pharmaceutical manufacturing is closely tied to its ability to deliver high-quality injectables at scale supported by strong regulatory frameworks and advanced manufacturing infrastructure. Partnering for Precision: Kilitch Healthcare’s Manufacturing Approach Within India’s evolving pharmaceutical landscape, Kilitch Healthcare (KHIL) has established itself as a dependable partner for companies seeking technically advanced, compliance-driven manufacturing solutions. With dedicated sterile facilities and a strong commitment to quality systems, KHIL supports pharmaceutical organizations across domestic and international markets. By aligning manufacturing processes with global regulatory expectations and investing in controlled environments, validated workflows, and trained technical teams, Kilitch Healthcare enables partners to achieve: This approach positions KHIL as a strategic collaborator in pharmaceutical contract manufacturing, particularly for organizations that prioritize precision, reliability, and long-term supply assurance. Strategic Implications for Pharmaceutical Decision-Makers For quality leaders, regulatory professionals, and operations executives, sterile manufacturing decisions have far-reaching consequences. The right
Sterile Injectable Manufacturing Services

GMP-Compliant Contract Manufacturing for Global Pharmaceutical Markets Sterile injectable products play a critical role in modern healthcare, particularly in acute care, critical therapies, and hospital-based treatments. As regulatory expectations increase and product complexity grows, pharmaceutical companies require manufacturing partners with advanced infrastructure, robust quality systems, and proven compliance capabilities. Kilitch Healthcare (KHIL) offers sterile injectable manufacturing services designed to meet the stringent demands of the global pharmaceutical industry. Our GMP-compliant facilities support scalable, reliable, and high-quality pharmaceutical manufacturing for companies seeking dependable contract manufacturing solutions in India and international markets. The Importance of Sterile Injectable Manufacturing in the Pharmaceutical Industry Sterile injectables represent one of the most regulated segments of pharmaceutical manufacturing. These products require controlled environments, validated aseptic processes, and strict adherence to GMP in the pharmaceutical industry to ensure patient safety and therapeutic efficacy. For pharmaceutical companies, outsourcing sterile manufacturing to a qualified partner enables access to specialized infrastructure while minimizing operational risk. A reliable sterile manufacturing partner ensures consistency, regulatory readiness, and supply continuity key requirements for hospital procurement teams and global healthcare partners. Kilitch Healthcare’s Sterile Manufacturing Capabilities Advanced Sterile Manufacturing Infrastructure Kilitch Healthcare operates dedicated sterile manufacturing facilities designed to support injectable dosage forms under controlled aseptic conditions. Our infrastructure is built to align with international regulatory expectations and good manufacturing processes, ensuring high standards of quality and operational integrity. Key facility features include: These capabilities enable KHIL to support pharmaceutical companies across multiple therapeutic segments while maintaining consistency and compliance. End-to-End Sterile Injectable Manufacturing Workflow Process Design and Technology Transfer Each project begins with a structured technology transfer process, ensuring seamless integration of formulations, specifications, and manufacturing requirements. Our technical teams work closely with pharmaceutical companies’ research and development functions to ensure process reproducibility and scalability. This collaborative approach supports pharmaceutical companies’ research and development objectives while maintaining regulatory alignment throughout the product lifecycle. Aseptic Filling and Sterile Processing Sterile injectables are manufactured using validated aseptic processes designed to minimize contamination risks. Our sterile manufacturing workflow incorporates: These measures support reliable pharmaceutical manufacturing outcomes, particularly for hospital-grade and export-oriented products. Quality Control and Batch Release Comprehensive quality control testing is conducted at every stage of production. This includes physical, chemical, and microbiological testing in accordance with approved specifications and regulatory guidelines. Only products that meet predefined quality standards are released, ensuring confidence for pharmaceutical companies, distributors, and healthcare institutions. Compliance with Global GMP Standards Regulatory Alignment and Quality Systems Kilitch Healthcare’s manufacturing operations are governed by robust quality management systems aligned with GMP in the pharmaceutical industry. Our facilities follow structured documentation, validation, and audit processes to support compliance across regulated and semi-regulated markets. Key compliance elements include: This compliance-driven approach positions KHIL among reliable pharmaceutical manufacturing companies in India. Business Advantages of Partnering with Kilitch Healthcare Scalable Manufacturing Capacity Our sterile manufacturing facilities are designed to support both small-batch and large-scale commercial production. This scalability allows pharmaceutical companies to respond to evolving market demands without additional capital investment. Operational Efficiency and Risk Mitigation By outsourcing sterile injectables manufacturing, pharmaceutical companies reduce the complexity associated with operating high-risk sterile facilities. KHIL manages manufacturing execution, compliance oversight, and quality assurance allowing partners to focus on commercialization and market expansion. Reliable Supply for Institutional and Export Markets Consistency and supply reliability are essential for hospital procurement teams and global distributors. Our structured production planning and validated processes ensure predictable supply chains and batch-to-batch consistency. Supporting India’s Role in Global Pharmaceutical Manufacturing India is recognized as a leading hub for pharmaceutical manufacturing, supported by a strong regulatory framework, skilled technical talent, and cost-efficient operations. As one of the best pharmaceutical companies in India offering contract manufacturing services, Kilitch Healthcare contributes to India’s reputation as a trusted global pharmaceutical partner. Our sterile manufacturing services support pharmaceutical companies operating as: Why Kilitch Healthcare Is a Trusted Sterile Manufacturing Partner Kilitch Healthcare combines technical expertise, regulatory discipline, and manufacturing reliability to support long-term partnerships. Our approach emphasizes: These principles allow KHIL to function not just as a manufacturer, but as a strategic partner to pharmaceutical companies across global markets. Applications Across Therapeutic and Institutional Segments Our sterile injectable manufacturing services support a wide range of applications, including: This versatility makes KHIL a preferred partner for pharmaceutical companies seeking dependable sterile manufacturing solutions. Conclusion: Reliable Sterile Injectable Manufacturing for Global Healthcare Sterile injectable manufacturing demands precision, compliance, and technical excellence. Kilitch Healthcare’s GMP-compliant facilities, structured workflows, and quality-focused culture provide pharmaceutical companies with a dependable manufacturing foundation. By partnering with KHIL, pharmaceutical manufacturers gain access to scalable sterile manufacturing capabilities aligned with global regulatory expectations supporting growth, reliability, and long-term success in the pharmaceutical industry. Contact Kilitch Healthcare For pharmaceutical companies, healthcare brands, and global partners seeking a reliable sterile injectable contract manufacturing partner, Kilitch Healthcare offers the expertise, infrastructure, and compliance assurance required for today’s healthcare markets. Connect with our team to discuss sterile injectable manufacturing requirements and explore partnership opportunities.
How Preservative-Free Eye Drops Are Transforming Modern Eye Care

The Importance of Eye Nutrition for Children in the Digital Age

The Evolution of Sterile Injectables in Healthcare

31st Dec 2022
Advancements in Ophthalmic Solutions: What You Need to Know

Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. sed do eiusmod tempor incididunt ut labore et dolore. Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. sed do eiusmod tempor incididunt ut labore et dolore. eiusmod tempor incididunt ut labore et dolore. Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed . dolore magna aliqua. sed do eiusmod tempor incididunt ut labore et dolore. eiusmod tempor incididunt ut labore et dolore. Lorem ipsum dolor sit amet, consectetur adipiscing elit, seddolore magna aliqua. sed do eiusmod tempor incididunt ut labore et dolore. eiusmod tempor incididunt ut labore et dolore. Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed
Leading the Way in Pharmaceutical Excellence: Kilitch Healthcare

